Sulfate ion bonding7/5/2023  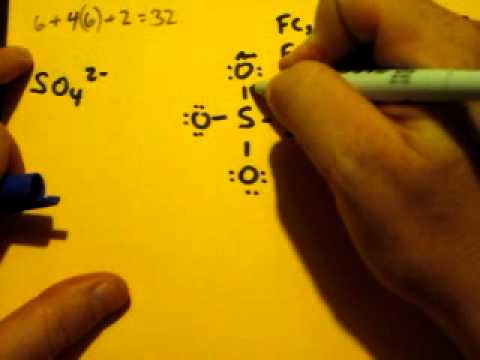
In electricity as the copper sulfate or in water treatment as the barium sulfate. They are largely used in constructions as the Epsom and gypsum salts. 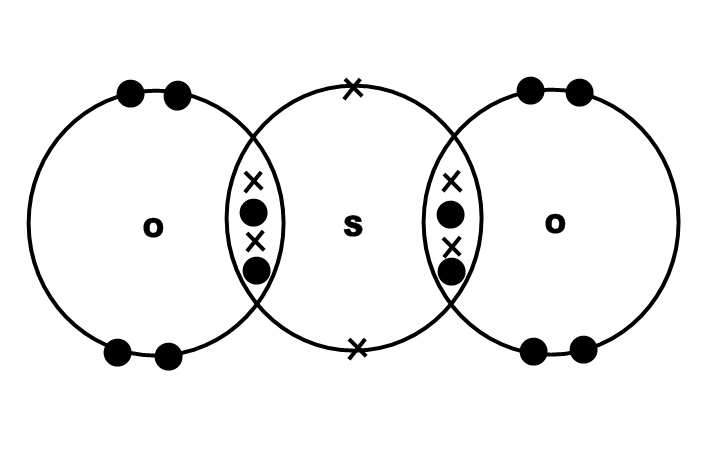
Uses: Sulfates are present in many natural compounds, for example in rocks, minerals and salts. Thus, the sulfate ion have 6 bonds instead of the typical 4 bonds for smaller atoms. On the other hand, the C-O bond is formed by a sigma bond. Most of them are soluble in water, except barium, calcium, lead, radium, strontium and mercury sulfates.Ĭhemical properties: Sulfate ion is formed due to the sulfur atom has an hypervalency, the S=O bonds are formed by a sigma and π bonds. In general they are solid, mostly powders. Physical properties: Sulfates vary in form according to the salt they are forming. For example, the preparation of metalic sulfates is made from the reaction of metalic salt (in this case barium chlorate) or the pure metal and sulfuric acid:īa(ClO 3) 2 H 2SO 4 → 2 HClO 3 BaSO 4 Preparation: Sulfates can be prepared with a wide variety of processes and depending on the metal to which sulfate is bond. Occurrence: Sulfate ion is largely found in nature as forming different salts and minerals with sodium, calcium, potassium and others. Its chemical structure can be written as below, in the common representations used for organic molecules. The geometry of the anion is tetrahedral. 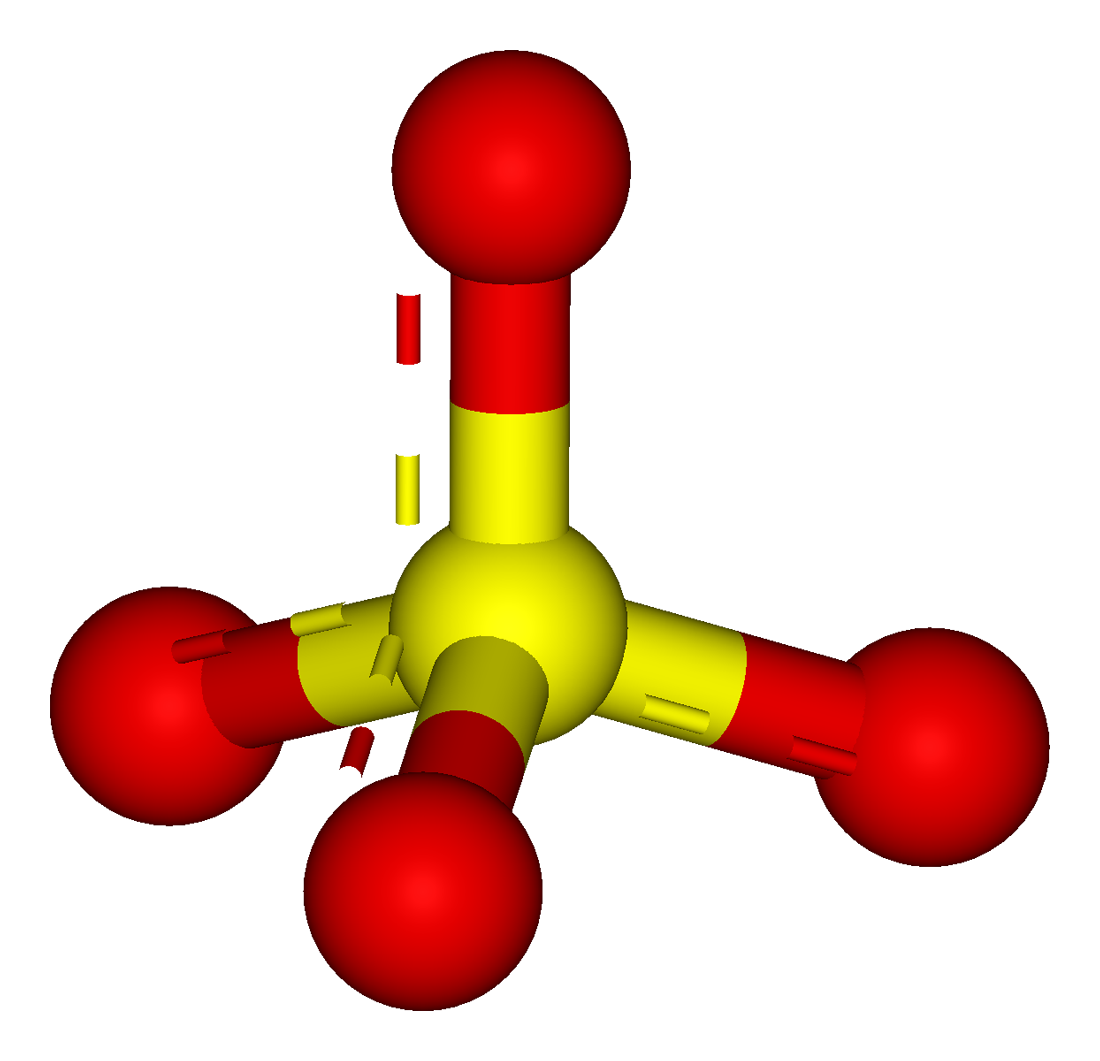
These last oxygen atoms are the responsible for the negative charge (-2) of the anion. This salt is formed by one sulfate center to which 4 atoms of oxygen are attached, 2 of these atoms are forming S=O bonds and the other two are S-O - bonds. In fact, it often is much more stable than uncharged molecules.Formula and structure: The sulfate ion formula is SO4 2- and the molar mass is 96.06 g mol -1. The sulfate ion is very stable: just because something is an ion does not mean it is unstable. In formal charge theory, the idea is indeed to try to keep individual FCs as close to zero as possible, but also to break the octet rule as little as possible. This gives it a total of 12 valence electrons. But the main reason is that sulfur only has 6 valence electrons in the first place, so it can only form up to 6 covalent bonds. It can be calculated (and has been, somewhere on this site that I cannot locate at the moment) that the participation of d orbitals in such compounds is very low – certainly far lower than an $\mathrm$, then the sulfur would have a total of 16 electrons in its valence shell, which would make it more unstable. However, their energy is too high for any meaningful bond to be formed. These orbitals are virtual (unoccupied) but do exist mathematically. Historically, the depiction of sulphate that you gave was ‘explained’ by sulphur using its 3d orbitals for bonding. Sulphur only has four such orbitals available (one 3s and three 3p) for bonding, so any electron exceeding the first eight would have to be placed in an antibonding orbital – but that would cause a decrease in bond order rather than an increase. 
On a very basic, general and simplified level, such a (localised) 2e2c bond is formed when an orbital of each atom overlap, resulting in a bonding and an antibonding orbital. Sulphur cannot form more than four traditional 2-electron-2-centre bonds (double bonds counting as two bonds, triple bonds as three bonds) due to a lack of available orbitals. If you come across any depiction of a main-group compound in which an atom has more electrons in its vicinity than the octet rule allows, chances are this depiction is either simplifying (for example: drawing a 4-electron-3-centre bond as if it were two single bonds), an unstable reaction intermediate or outright incorrect. In this structure, sulphur is surrounded by exactly eight valence electrons as predicted by the octet rule. Unfortunately, I found no calculation of sulphate’s structure in a quick search but rest assured that it will be the logical extension of sulphite. Martin performed a calculation on the closely related sulphite ion (in which there is one less oxygen leading to a lone pair on sulphur) which shows zero π-type bonding orbitals. The correct structure of sulphate, shown below, has exactly zero double bonds. What you have seen is not an accurate depiction of the bonding situation according to the currently accepted theory. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
